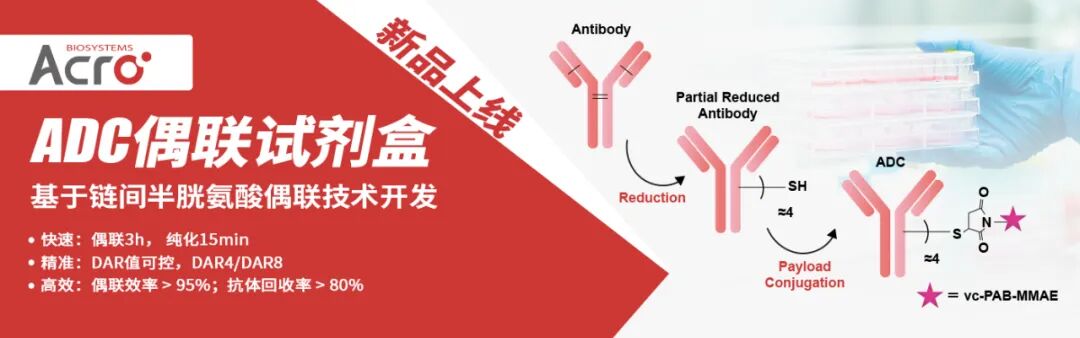
ADC(抗體藥物偶聯物)是 “抗體 - 連接子 - 細胞毒性載荷” 構成的靶向藥物,核心是通過偶聯技術將細胞毒性載荷(如MMAE、DXd)精準連接到抗體上,實現“靶向遞送毒性藥物至腫瘤細胞” 的效果 ——偶聯技術決定載荷如何高效、穩定地與抗體結合,直接影響 ADC 的藥效、安全性和生產可行性。
-
偶聯流程漫長,動輒耗費數天甚至更久,嚴重拖慢研發節奏?
-
DAR值像“開盲盒”,難以精確控制,導致產物異質性高,影響藥效評估?
-
抗體活性和回收率在偶聯過程中損耗嚴重,寶貴樣品令人心疼?
ACROBiosystems百普賽斯全新推出的抗體偶聯試劑盒,為您帶來“快、準、穩、簡” 的ADC制備體驗,保證抗體活性與高回收率,得到的ADC產物可用于細胞或動物實驗,以篩選合適的候選ADC藥物。
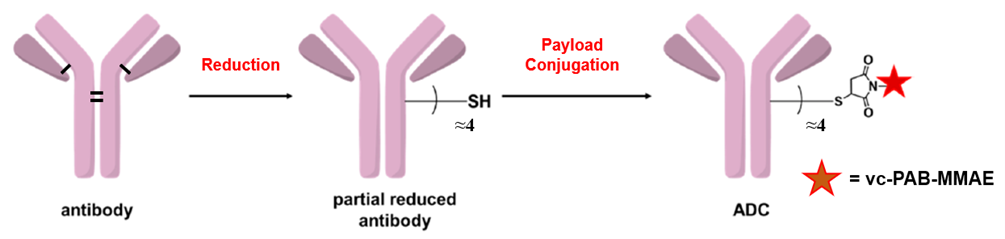
本試劑盒基于鏈間半胱氨酸偶聯技術開發:抗體的鏈間二硫鍵可以被還原產生內源性鏈間半胱氨酸殘基,具有馬來酰亞胺官能團的有效載荷藥物可以特異性偶聯這些半胱氨酸殘基并形成結構穩定的ADC。依靠這種經典的偶聯策略,本試劑盒能夠實現快速可控的ADC制備,只需要3個小時即可獲得平均藥物抗比率(DAR)為4.0±0.5的ADC偶聯物,且獲得的ADC偶聯物表現出顯著的細胞毒性作用,為ADC藥物篩選和機制研究提供了可靠的實驗工具。
-
偶聯時間短:3 小時內即可生成抗體藥物偶聯物(ADCs)
-
藥物抗體比率(DAR)值可控:可精準調控 DAR,同時保留良好抗體活性
-
偶聯效率與回收率高:偶聯效率 > 95%,抗體回收率 > 80%
-
無需高濃度抗體:僅需 2 mg/mL 的抗體濃度即可實現反應
-
純化簡單:使用離心脫鹽柱,15 分鐘內即可完成純化
|
貨號 |
產品名稱 |
|
ADC Conjugation Kit (MMAE, DAR4, 200ug, for human IgG1) |
|
|
ADC Conjugation Kit (MMAE, DAR4, 1mg, for human IgG1) |
|
|
ADC Conjugation Kit (MMAF, DAR4, 200ug, for human IgG1) |
|
|
ADC Conjugation Kit (MMAF, DAR4, 1mg, for human IgG1) |
|
|
ADC Conjugation Kit (Exatecan, DAR8, 200ug, for human IgG1) |
|
|
ADC Conjugation Kit (Exatecan, DAR8, 1mg, for human IgG1) |
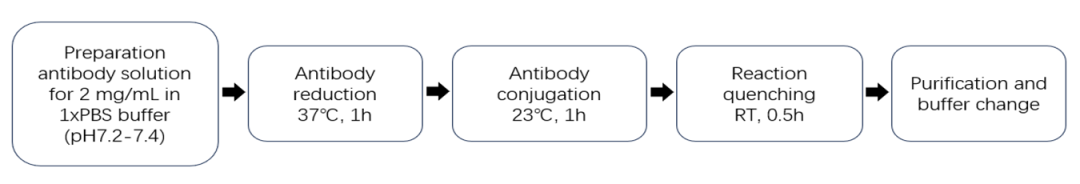
試劑盒組分(以MMAE, DAR4, 貨號:ADC-P013為例):

以下ADC產品使用ADC偶聯試劑盒(MMAE, DAR4, 貨號:ADC-P013)制備。
抗體信息:曲妥珠單抗生物類似物。
?DAR值及純度經HPLC驗證 (HIC+SEC):
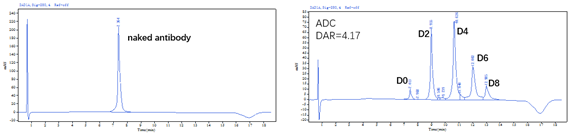
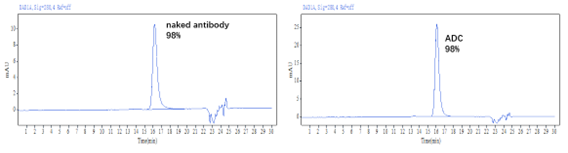
Figure 1. The ADC was prepared using the ADC Conjugation Kit (MMAE, DAR4) and analyzed by HIC and SEC-HPLC. The average drug-antibody ratio (DAR) is 4.0±0.5, and the purity of the ADC is greater than 95%.
?DAR值經質譜驗證
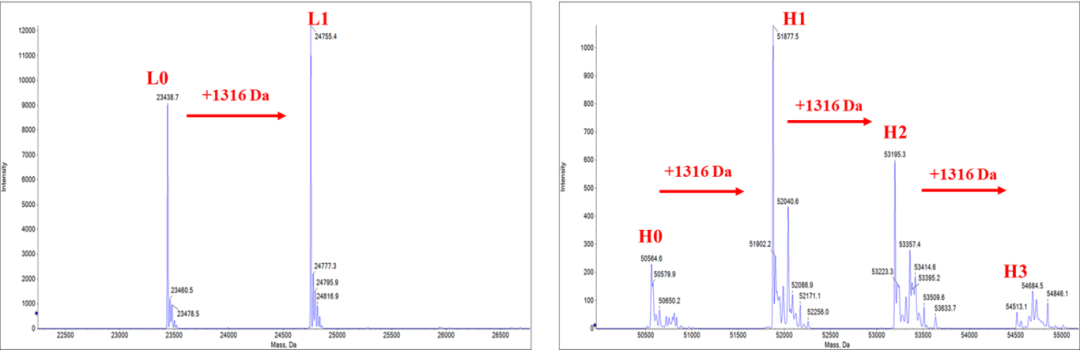
Figure 2. The DAR (3.82) was calculated from the weighted average of the deconvoluted MS peak areas using LC-MS/MS. The results showed the deconvoluted mass spectra of light chains and heavy chains, and the increase in molecular weight caused by the coupling payload (1316±3 Da). The heterogeneity in N-glycosylation of heavy chain adds to the complexity of the mass spectrum.
?抗原結合能力檢測
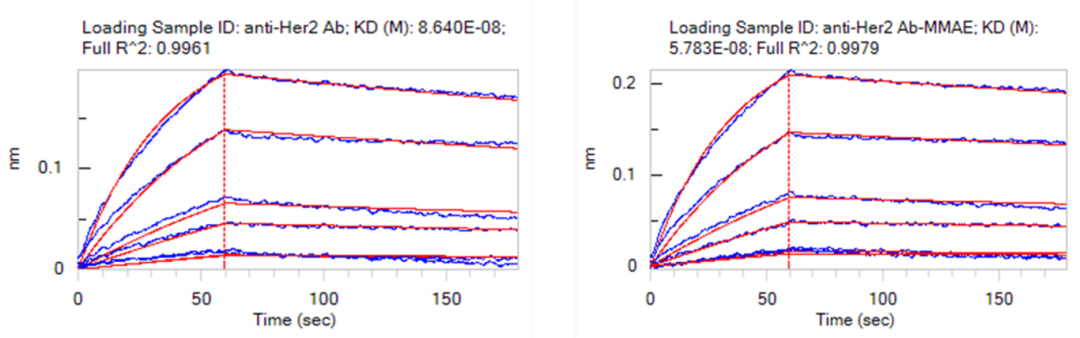
Figure 3. Binding affinity of anti-Her2 antibody and anti-Her2 antibody–MMAE conjugate to human Her2 (Cat. No. HE2-H5225) as determined by BLI (Bio-Layer Interferometry). The conjugate exhibits nanomolar affinity (0.57 nM) for human Her2, comparable to the naked antibody (0.86 nM).
?體外細胞毒性驗證
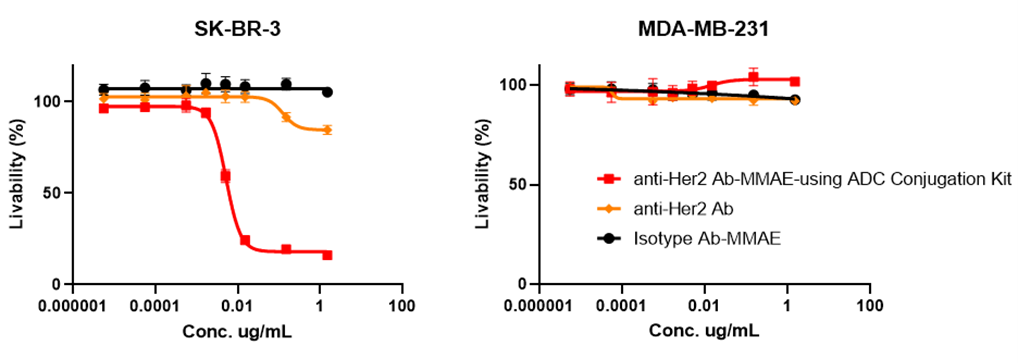
Figure 4. In vitro cytotoxicity assays: The ADC can bind and internalize in target cells (SK-BR-3) with high expression of HER2 and release MMAE inside the cells to induce a cytotoxic effect (IC50=0.0058 µg/mL). Meanwhile, no cytotoxicity was observed in HER2 receptor-nagetive cell lines (MDA-MB-231).
?不同規格Kit一致性驗證

Figure 5. In vitro cytotoxicity of products prepared by different size of the ADC Conjugation Kit (Cat. No. ADC-P013, ADC-P014). The result shows very high consistency (RSD<10%).
?穩定性驗證
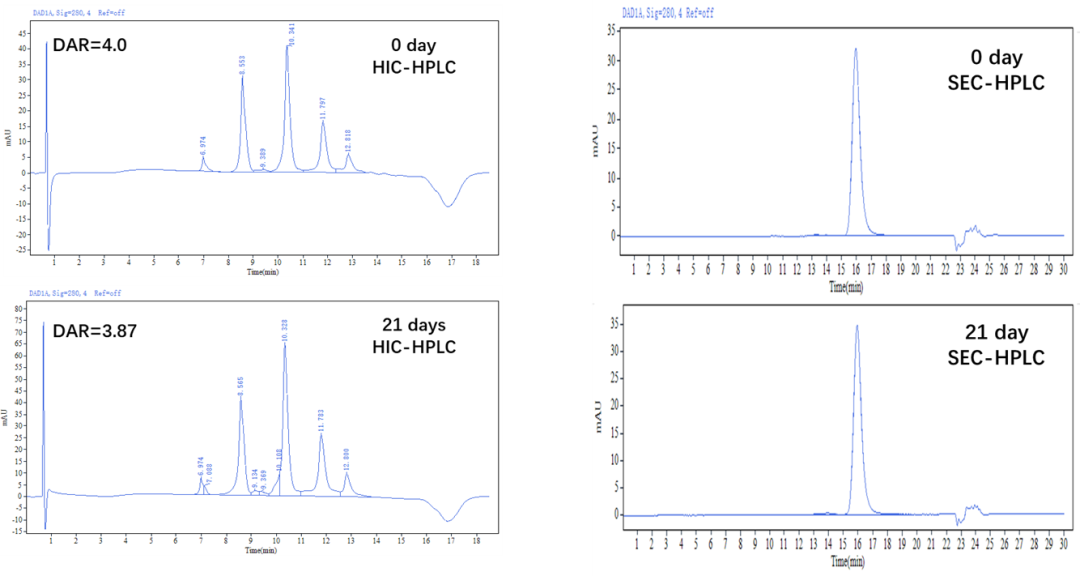
Figure 6. The MMAE-ADC was prepared using the ADC Conjugation Kit, which were left at 37℃ for 21 days. The ADCs were analyzed by HIC and SEC-HPLC. The average drug-antibody ratio (DAR) is 4.0±0.5, and the purity of the ADC is greater than 95%.

除此之外,ACROBiosystems百普賽斯推出了AGLink®新型DAR2&4定點偶聯試劑盒,該試劑盒基于YTConju™糖基偶聯技術平臺開發,包含毒素(MMAE)、反應官能團(Tz/DBCO)、檢測標簽(Biotin)三大模塊,為偶聯藥物的早期研究及相關生物學實驗提供強大支持。


ACROBiosystems
inquiry@acrobiosystems.com
15117918562
(備注:姓名+公司)
